Rhizosphere Microbial Community Structure of Transgenic versus Non-Transgenic Maize Varieties
Keywords:
Rhizosphere , Diversity , Community , TransgenicAbstract
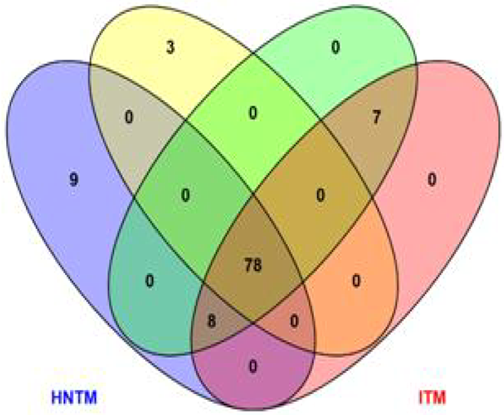
The soil rhizosphere community is an important indicator of the overall health of the plant, and the resulting yield at harvest. The study determined the rhizosphere microbial community structure of transgenic and non-transgenic maize plants. A farm plot was divided into 4 sections for transgenic maize (SAMMAZ 74T) and non-transgenic variety 8325-8. The maize was infected at the tasseling stage and phenolic content determined after 10 days. The microbial community indices were subsequently determined using metagenomic tools. The physicochemical parameters and microbial population of the soil for maize 8325-8 and SAMMAZ 74T were not significantly differently at P<0.05. The soil with infected maize were the most diverse with Shannon index 1.834. The soil with infected maize were the most diverse with Shannon index 1.834. The transgenic maize SAMMAZ 74T had the highest dominance of bacteria (0.2536) and fungi (0.10930). Proteobacteria was the predominant bacteria phylum while Ascomycota was the predominant fungal phylum. The study concludes that the non-transgenic maize had better contribution to the soil microbial community than the transgenic maize variety.
Published
How to Cite
Issue
Section
Copyright (c) 2026 Olukayode Olugbenga Orole, Oluwatosin Okunade, Bashir Sani, Grace Kwala, Stella Ladi Ageba, Timothy Adejumo (Author)

This work is licensed under a Creative Commons Attribution-ShareAlike 4.0 International License.
How to Cite
Most read articles by the same author(s)
- Peter Uteh Upla, Bashiru Eya Sani, Osuyi Gerard Uyi, Igoche Naomi Ibe, Salamatu Idris, Oluwatosin Okunade, Naja'atu Shehu Hadi, Abdulaziz Aliyu Dardau, Kabiru Shuaibu, Fatima Yusuf Al-Mustapha, Gladys Abel Angbalaga, Hand Carriage of Microorganisms by Students of Federal University of Lafia, Nasarawa State, Nigeria , Lafia Journal of Scientific and Industrial Research: Volume 1, Issue 1 & 2 (October, 2023), Lafia Journal of Scientific and Industrial Research (LJSIR)
- Stella Ladi Ageba, Olukayode Olugbenga Orole, Gerard Osuyi Uyi, Maryam Hassan Muhammad, Femi Gbadeyan, Characterisation of Antibiotic Resistant Pseudomonas Species in Environmental and Clinical Samples in Lafia, Nasarawa State , Lafia Journal of Scientific and Industrial Research: Volume 4, Issue 1 (April, 2026), Lafia Journal of Scientific and Industrial Research (LJSIR)
- Olukayode Olugbenga Orole, Henry David Dimut, Femi Gbadeyan, Phylogenetic Characterisation of Antimycotoxigenic Fungal and Bacterial Endophytes from Maize Roots , Lafia Journal of Scientific and Industrial Research: Volume 4, Issue 1 (April, 2026), Lafia Journal of Scientific and Industrial Research (LJSIR)
Similar Articles
- Collina Kambai, Danjuma Filibus Dami, Adams Adamanyiwa Chaskda, Bird Composition, Abundance and Diversity across Habitat Types in Shere Hills Reserve, Plateau State, Nigeria , Lafia Journal of Scientific and Industrial Research: Volume 3, Issue 1 (April, 2025), Lafia Journal of Scientific and Industrial Research (LJSIR)
- Osuyi Gerard Uyi, Bashiru Eya Sani, Olukayode Orole, Gift Chidiebere Ejindu, Afamefuna Okafor Dunku, Aleruchi Chuku, Peter Uteh Upla, Isolation and Characterization of Phosphorus Solubilizing Bacteria from Millet Rhizosphere in Lafia metropolis , Lafia Journal of Scientific and Industrial Research: Volume 2, Issue 1 (April, 2024), Lafia Journal of Scientific and Industrial Research (LJSIR)
- Alhassan U. M., Kana H. A., Zainab H., Phenotypic diversity and performance for some agronomic characters in sesame (Sesamum indicum L.) germplasm collection from Nasarawa state, Nigeria , Lafia Journal of Scientific and Industrial Research: Volume 3, Issue 1 (April, 2025), Lafia Journal of Scientific and Industrial Research (LJSIR)
- Osuyi Gerard Uyi, Olabisi Peter Abioye, Jeremiah Bala, Philip Alkali Tsado, Sudesh Kumar, Syauqi Ahmad, Isolation and Optimization of Phosphorus Solubilizing Bacteria from Maize Rhizosphere in Nasarawa State, Nigeria , Lafia Journal of Scientific and Industrial Research: Volume 1, Issue 1 & 2 (October, 2023), Lafia Journal of Scientific and Industrial Research (LJSIR)
- Jude Chinedu Onwuka, Daniel Apeh Egyegbola, Bako Danjuma, Amos Idzi Ambo, Bilyamin Ishaq, Enebi Estella Jasper, Gladys Nkeiruka Onwuka, Lucy Ooja Agho, Adamu Usman, Agu Matthew Onyema, Ecological Health Risk Evaluation of Potentially Toxic Metals in Saline Soils Used for Table Salt Production in Awe Salt Mining Community of Nasarawa State , Lafia Journal of Scientific and Industrial Research: Volume 4, Issue 1 (April, 2026), Lafia Journal of Scientific and Industrial Research (LJSIR)
- Oluwaseun Adesoye, Tolulope Oyeniyi, Ayodele Babalola, Olalekan Olagundoye, Romoke Izekor, Oluwakemi Adetunji, Israel Akinsete, Adeniyi Kamoru, Callistus Akinleye, Adewale Adediran, Chidinma Isaac, Adedapo Adeogun, Entomological Collections and Identifications of Mosquito Faunas in Selected Area Councils of Nigeria Federal Capital Territory , Lafia Journal of Scientific and Industrial Research: Volume 2, Issue 2 (October, 2024), Lafia Journal of Scientific and Industrial Research (LJSIR)
- Oludare Agboola, Evangeline Abah, Samuel Ijimbili, Oche Andrew, Irene Unazi, Audud Onyemocho, Adelanwa E., Assessment of Waste Management Practices among Residents of Benue South , Lafia Journal of Scientific and Industrial Research: Volume 2, Issue 1 (April, 2024), Lafia Journal of Scientific and Industrial Research (LJSIR)
- Gbenga Ogunsanwo, Phillip T. Odulaja, A. A. Omotunde, Olakunle O. Solanke, Air Quality Index prediction using Deep learning for Lagos State in Nigeria , Lafia Journal of Scientific and Industrial Research: Volume 3, Issue 1 (April, 2025), Lafia Journal of Scientific and Industrial Research (LJSIR)
- Dantani Dauda Odonye, Joseph F. Nfongeh , Femi A. Gbadeyan, Adamu Abisabo, Shuaibu U. Okposhi , Enoch P. Odonye , Oluwatosin A. Okunade, Antibiotic Resistant Patterns of Salmonella Serovars Isolated from Clinical Stool Samples in Nasarawa State, Nigeria , Lafia Journal of Scientific and Industrial Research: Volume 4, Issue 1 (April, 2026), Lafia Journal of Scientific and Industrial Research (LJSIR)
- Ajah Precious Amarachi , Ogbuta Philip Kelechukwu, Kalu Patience Ogbuta, Ajah Rosemary Ogechukwu, Afolabi Oluwatuyi Samson, Assessment of the Perception of Malaria Prevention among Pregnant Women Attending Antenatal Clinic at Obiozara Health Center Uburu, Ohaozara, Ebonyi State , Lafia Journal of Scientific and Industrial Research: Volume 4, Issue 1 (April, 2026), Lafia Journal of Scientific and Industrial Research (LJSIR)
You may also start an advanced similarity search for this article.




