Characterisation of Antibiotic Resistant Pseudomonas Species in Environmental and Clinical Samples in Lafia, Nasarawa State
DOI :
https://doi.org/10.62050/ljsir2026.v4n1.824Mots-clés :
Pseudomonas, antibiotic resistance, multidrug resistance, environmentRésumé
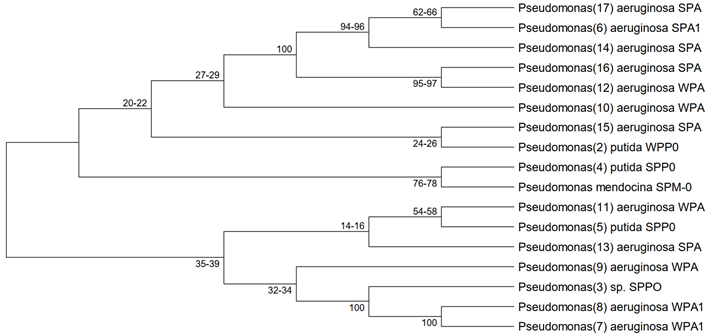
Soil, water, and animals are reservoirs for resistance genes and multidrug resistant bacteria. The genus Pseudomonas is widely distributed in nature, with Pseudomonas aeruginosa being the most clinically significant. The aim of the study was to characterize antibiotic resistance in Pseudomonas species in environmental and clinical samples in Lafia, Nasarawa State. Water and soil (50 each) samples were collected five times per week, along with 50 urine samples. Samples with significant bacteriuria were analysed for positive isolates. The presence of virulence and antibiotic resistance genes on their DNA was confirmed with PCR analysis. Water samples had Pseudomonas counts ranging from 1.0x05 to 17.0x105 CFU/mL, and soil samples had highest count of 14.0x104 CFU/g. Pseudomonas species were 86.7% resistant to Augmentin and 75% resistant to Ceftazidime, while Ofloxacin had the lowest resistant isolates at 25%. The multidrug resistance was 72.7% in the water isolates, 72.2% in urine, and 60.0% in soil isolates as the lowest. There was no significant association between sample source and MDR prevalence (p > 0.05). The resistance gene bla_VIM and virulence gene pvdM was each carried by 14 (82.4%) of the Pseudomonas sp. and six of them co-carried bla_VIM and bla_NDM genes. Pseudomonas mendocina isolate haboured neither virulence nor resistance genes. The findings calls for concern as the genes haboured by the Pseudomonas species encode resistance to carbapenems, the last resort antibiotics. Pseudomonas putida, a non-pathogenic isolates haboured bla_VIM and bla_NDM genes making them a reservoir for horizontal transfer to other isolates and bacteria in the environment.
##plugins.themes.default.displayStats.downloads##
Références
Murray, C. J. L., Ikuta, K. S., Sharara, F., Swetschinski, L., Aguilar, G. R., Gray, A., Han, C., Bisignano, C., Rao, P., Wool, E., Johnson, S. C., Browne, A. J., Chipeta, M. G., Fell, F., Hackett, S., Haines-Woodhouse, G., Hamadani, B. H. K., Kumaran, E. A. P., McManigal, B., Naghavi, M. (2022). Global burden of bacterial antimicrobial resistance in 2019: A systematic analysis. The Lancet, 399(10325), 629–655. https://doi.org/10.1016/S0140-6736(21)02724-0
Singh, C. K., & Kaur Sodhi, K. (2023). The emerging significance of nanomedicine-based approaches to fighting COVID-19 variants of concern: a perspective on the nanotechnology’s role in COVID-19 diagnosis and treatment. Frontier Nanotechnology. 4, 1–13. January. https://doi.org/10.3389/fnano.2023.1084033
Carmo, L.P.; Nielsen, L.R.; Alban, L.; da Costa, P.M.; Schüpbach-Regula, G.; & Magouras, I. (2018). Veterinary Expert Opinion on Potential Drivers and Opportunities for Changing Antimicrobial Usage Practices in Livestock in Denmark, Portugal, and Switzerland. Frontier Veterinary Science. 5, 29.
Fernandez, B., María, A. E., Llambías, C., Jordana-Lluch, E., Oliver, A., & Macià, M. D. (2023). Mechanisms of antibiotic resistance in Pseudomonas aeruginosa biofilms. Biofilm. 5, 100129. https://doi.org/10.1016/j.bioflm.2023.100129
Biondo, C. (2023). Bacterial antibiotic resistance: the most critical pathogens. Pathogens 12 (1), 116–214. https://doi.org/10.3390/pathogens12010116
Elfadadny, A., Soliman, A. M., & Ramadan, H. Antimicrobial resistance mechanisms in Pseudomonas aeruginosa: Recent trends and clinical impact. Frontiers Microbiology, 15, 1178902. https://doi.org/10.3389/fmicb.2024.1178902
Darby, E. M., Trampari, E., Siasat, P., Solsona Gaya, M., Alav I., & Webber, M. A.(2023). Molecular mechanisms of antibiotic resistance revisited. Nature Reviews Microbiology. 21 (5), 280–295. https://doi.org/10.1038/s41579-022-00820
Awanye, A. M., Ibezim, C. N., Stanley, C. N., Onah, H., Okonko, I. O., & Egbe, N. E. (2022). Multidrug-resistant and extremely drug-resistant Pseudomonas aeruginosa in clinical samples from a tertiary healthcare facility in Nigeria. Turkish Journal of Pharmaceutical Sciences, 19(4), 447–454. https://doi.org/10.4274/tjps.galenos.2021.66066
Adekunle, C., Mustapha, A., & Odewale G. (2021). Detection of antibiotic resistance genes among multiple drug resistant Pseudomonas aeruginosa isolated from clinical sources in selected health institutions in Kwara state. Infect Disord Drug Targets; 21: e170721187999.2021
Anwar, M., Iqbal, Q., & Saleem, F. (2020). Improper disposal of unused antibiotics: an often overlooked driver of antimicrobial resistance. Expert Revised Anti-Infective Therapy 18 (8), 697–699. https://doi.org/10.1080/14787210.2020.1754797
Adesoji, A. T., Onuh, J. P., Palang, I. P., Liadi, A. M., & Musa, S. (2023). Prevalence of multi-drug resistant Pseudomonas aeruginosa isolated from selected residential sewages in Dutsin-Ma, Katsina State, Nigeria. Journal of Public Health in Africa, 14(2), 2152. https://doi.org/10.4081/jphia.2023.2152
Owoseni, M. C., Oyigye, O., Sani, B., Lamin, J., & Chere, A. (2022). Antimicrobial resistance and virulence genes profiling of Proteus species from poultry farms in Lafia, Nigeria. African Journal of Medical and Pharmaceutical Sciences, 5(2). ojshostng.com
Arowolo, M. T., Orababa, O. Q., Olaitan, M. O., Osibeluwo, B. V., Essiet, U. U., Batholomew, O. H., Ogunrinde, O. G., Lagoke, O. A., Soriwei, J. D., Ishola, O. D., Ezeani, O. M., Onishile, A. O., & Olumodeji, E. (2023). Prevalence of carbapenem resistance in Acinetobacter baumannii and Pseudomonas aeruginosa in sub-Saharan Africa: A systematic review and meta-analysis. PLOS ONE, 18(11), e0287762. https://doi.org/10.1371/journal.pone.0287762
Adesakin, T. A., Oyewale, A. T., Bayero, U., Mohammed, A. N., Aduwo, I. A., Ahmed, P. Z., Abubakar, N. D., & Barje, I. B. (2020). Assessment of bacteriological quality and physico-chemical parameters of domestic water sources in Samaru community, Zaria, Northwest Nigeria. Heliyon, 6(8), e04773. https://doi.org/10.1016/j.heliyon.2020.e04773
Li, Y., Qu, Y., Yang, H., Zhou, X., Xiao, P., & Shao, T. (2023). Combatting biofilms in potable water systems: A comprehensive overview to ensuring industrial water safety. Environmental Microbiology Reports, 15(6), 445. https://doi.org/10.1111/1758-2229.13207
Kelly-Quinn, M., Jennings, E., Antunes, P., & Fenton, O. (2019). The Environmental Impact of Cattle Access to Watercourses: A Review. Journal of Environmental Quality, 48(2), 340-351. https://doi.org/10.2134/jeq2018.04.0167
Akhtar, N., Syakir Ishak, M. I., Bhawani, S. A., & Umar, K. (2020). Various Natural and Anthropogenic Factors Responsible for Water Quality Degradation: A Review. Water, 13(19), 2660. https://doi.org/10.3390/w13192660
Mohammed, K., & Abdullahi, M. (2025). Molecular detection and resistance profile of Pseudomonas aeruginosa and Escherichia coli isolated from pediatric patients in selected hospitals in Sokoto Metropolis. African Journal of Advances in Science and Technology Research, 20(1), 15–31. https://doi.org/10.62154/ajastr.2025.020.01012
Anju, V. T., Busi, S., Imchen, M., Kumavath, R., Mohan, M. S., Salim, S. A., Subhaswaraj, P., & Dyavaiah, M. (2022). Polymicrobial Infections and Biofilms: Clinical Significance and Eradication Strategies. Antibiotics, 11(12), 1731. https://doi.org/10.3390/antibiotics11121731
Kakoullis, L., Papachristodoulou, E., Chra, P., & Panos, G. (2021). Mechanisms of Antibiotic Resistance in Important Gram-Positive and Gram-Negative Pathogens and Novel Antibiotic Solutions. Antibiotics, 10(4), 415. https://doi.org/10.3390/antibiotics10040415
Aggarwal, A., Bhalla, M., & Fatima, K. H. (2020). Detection of New Delhi metallo-beta-lactamase enzyme gene bla NDM-1 associated with the Int-1 gene in Gram-negative bacteria collected from the effluent treatment plant of a tuberculosis care hospital in Delhi, India. Access Microbiology, 2(6), acmi000125. https://doi.org/10.1099/acmi.0.000125
Qin, S., Xiao, W., Zhou, C., Pu, Q., Deng, X., Lan, L., Liang, H., Song, X., & Wu, M. (2022). Pseudomonas aeruginosa: Pathogenesis, virulence factors, antibiotic resistance, interaction with host, technology advances and emerging therapeutics. Signal Transduction and Targeted Therapy, 7(1), 199. https://doi.org/10.1038/s41392-022-01056-1
Yousefi-Avarvand, A., Khashei, R., Ebrahim-Saraie, H. S., Emami, A., Zomorodian, K., & Motamedifar, M. (2015). The Frequency of Exotoxin A and Exoenzymes S and U Genes among Clinical Isolates of Pseudomonas aeruginosa in Shiraz, Iran. International Journal of Molecular and Cellular Medicine, 4(3), 167. https://pmc.ncbi.nlm.nih.gov/articles/PMC4644528/
Adenipekun, E. O., Akinleye, E. F., Tewogbade, O. A., & Iwalokun, B. A. (2023). Detection of virulence genes and multidrug resistance in Pseudomonas aeruginosa clinical isolates from a public hospital in Lagos, Nigeria. Scientific African, 22, e01950. https://doi.org/10.1016/j.sciaf.2023.e01950
Téléchargements
Publiée
Numéro
Rubrique
Licence
(c) Copyright Stella Ladi Ageba, Olukayode Olugbenga Orole, Gerard Osuyi Uyi, Maryam Hassan Muhammad, Femi Gbadeyan (Author) 2026

Ce travail est disponible sous licence Creative Commons Attribution - Partage dans les Mêmes Conditions 4.0 International.









