Crude and Nano Enhanced Antifungal Properties of Acalypha wilkesiana leaf extract against Trichophyton mentagrophytes
الكلمات المفتاحية:
Array، Array، Array، Arrayالملخص
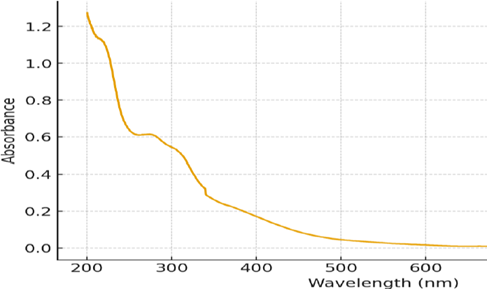
Trichophyton mentagrophytes is a dermatophyte fungus responsible for infections of keratinized tissues such as the skin, hair, and nails. This disease can be caused by poor hygiene, a warm environment, and an individual's weakened immune system. In this study, we examined the antifungal properties of crude and nano-enhanced Acalypha wilkesiana against T. mentagrophytes. Leaves of Acalypha wilkesiana were extracted with ethanol by maceration. Phytochemical analysis was conducted using conventional methods, and zinc oxide nanoparticles were prepared using a green synthesis technique. The Antifungal potential of A. wilkesiana was evaluated using the agar well diffusion test and microdilution method. The data were analyzed using two-way ANOVA, with a significance at P ≤ 0.001. The presence of alkaloids (5.82 ± 0.12 mg/g), flavonoids (8.35 ± 0.15 mg/g), and phenols (7.96 ± 0.21 mg/g) was noted during phytochemical screening. The gas chromatography-mass spectrometry (GC–MS) analysis confirmed ten compounds in the extract. Among the compounds, palmitic acid had the highest percentage peak (18.2%) with a molecular weight of 256 and a retention time of 5.43 minutes. The nano-enhanced formulation demonstrated greater inhibition zones than the crude formulation. Particularly, nano-formulated A. wilkesiana demonstrated significant improvement in antifungal properties against T. mentagrophytes (F = 1,261.50; P ≤ 0.001), with a zone of inhibition of 32.00 ± 1.00 mm compared to 23.00 ± 1.00 mm for the crude extract. Minimal Inhibitory Concentration (MIC) values showed a dose-response effect on the fungi.
منشور
كيفية الاقتباس
إصدار
القسم
الحقوق الفكرية (c) 2026 Ogbonnaya Nkechinyere Agwa, Owoseni Mojisola Christiana, Aleruchi Chuku, Nworie Chukwuemeka Richard (Author)

هذا العمل مرخص بموجب Creative Commons Attribution-ShareAlike 4.0 International License.
كيفية الاقتباس
الأعمال الأكثر قراءة لنفس المؤلف/المؤلفين
- Mojisola Christiana Owoseni, Bashiru Eya Sani, Hamida Damilare Usman, Seroprevalence and risk factors of Hepatitis B and C Virus infection Among Inmates in a Correctional Service, Nasarawa State, Nigeria , Lafia Journal of Scientific and Industrial Research: Volume 2, Issue 1 (April, 2024), Lafia Journal of Scientific and Industrial Research (LJSIR)
المؤلفات المشابهة
- Aleruchi Chuku, Osuyi Gerard Uyi, Godwin Attah Obande, Ayomide Hassan Labulo , Promise Ujunwa Ikechuku, Characterization and Deployment of Zinc Oxide Nanoparticles of Edible Mushroom with Antifungal Activity against Selected Dermatophytic Fungi , Lafia Journal of Scientific and Industrial Research: Volume 4, Issue 1 (April, 2026), Lafia Journal of Scientific and Industrial Research (LJSIR)
- Aleruchi Chuku, Bashiru Eya Sani, Pedro Akharenegbe, Godwin Attah Obande, Prevalence and Antifungal Susceptibility of Fungal Isolates Associated with Otomycotic Patients in Lafia , Lafia Journal of Scientific and Industrial Research: Volume 1, Issue 1 & 2 (October, 2023), Lafia Journal of Scientific and Industrial Research (LJSIR)
- Murshida Abdullahi Ganuwa, Pedro Akharenegbe, Hannah Eyo Nsemoh, Isah Ibrahim Okposhi, Olukayode Orole, Resistance to Azole Drugs by Fungal Species Isolated from Date Palm Samples within Lafia Metropolis, Nigeria , Lafia Journal of Scientific and Industrial Research: Volume 2, Issue 2 (October, 2024), Lafia Journal of Scientific and Industrial Research (LJSIR)
- Stella Ladi Ageba, Olukayode Olugbenga Orole, Gerard Osuyi Uyi, Maryam Hassan Muhammad, Femi Gbadeyan, Characterisation of Antibiotic Resistant Pseudomonas Species in Environmental and Clinical Samples in Lafia, Nasarawa State , Lafia Journal of Scientific and Industrial Research: Volume 4, Issue 1 (April, 2026), Lafia Journal of Scientific and Industrial Research (LJSIR)
- Precious Opeyemi Oghenetega , Nkechi Eucharia Egbe, Jeremiah Appah, Kingsley Onuh Chukwudi, Abba Umar Hassan, Determination of Antibiotic-Resistant Genes in Bacteria from Borehole Water Samples in Kaduna Metropolis , Lafia Journal of Scientific and Industrial Research: Volume 4, Issue 1 (April, 2026), Lafia Journal of Scientific and Industrial Research (LJSIR)
- L. Y. Adogo, K. M. Julia, P. D. Joshua, Occurrence and Antifungal Susceptibility of Candida Species Isolated from Pregnant Women in a Tertiary Hospital , Lafia Journal of Scientific and Industrial Research: Volume 1, Issue 1 & 2 (October, 2023), Lafia Journal of Scientific and Industrial Research (LJSIR)
- Dantani Dauda Odonye, Joseph F. Nfongeh , Femi A. Gbadeyan, Adamu Abisabo, Shuaibu U. Okposhi , Enoch P. Odonye , Oluwatosin A. Okunade, Antibiotic Resistant Patterns of Salmonella Serovars Isolated from Clinical Stool Samples in Nasarawa State, Nigeria , Lafia Journal of Scientific and Industrial Research: Volume 4, Issue 1 (April, 2026), Lafia Journal of Scientific and Industrial Research (LJSIR)
- Ayorinde O. Nejo, Ademola J. Adetona, Adetoun Lawal, Green Synthesis of Nickel Oxide Nanoparticles and its Application in the Degradation of Methyl Red , Lafia Journal of Scientific and Industrial Research: Volume 2, Issue 2 (October, 2024), Lafia Journal of Scientific and Industrial Research (LJSIR)
- Ebah Esther Eneyi, Emmanuel Olumuyiwa Onifade, Dim Chinemerem Ugochinyere, Augustine David Aondoackaa, Antibiotic Susceptibility Pattern of Salmonella typhi Isolated from Hostel Tap Water of a Tertiary Institution in Makurdi , Lafia Journal of Scientific and Industrial Research: Volume 2, Issue 2 (October, 2024), Lafia Journal of Scientific and Industrial Research (LJSIR)
- Olukayode Olugbenga Orole, Henry David Dimut, Femi Gbadeyan, Phylogenetic Characterisation of Antimycotoxigenic Fungal and Bacterial Endophytes from Maize Roots , Lafia Journal of Scientific and Industrial Research: Volume 4, Issue 1 (April, 2026), Lafia Journal of Scientific and Industrial Research (LJSIR)
يمكنك أيضاً إبدأ بحثاً متقدماً عن المشابهات لهذا المؤلَّف.




