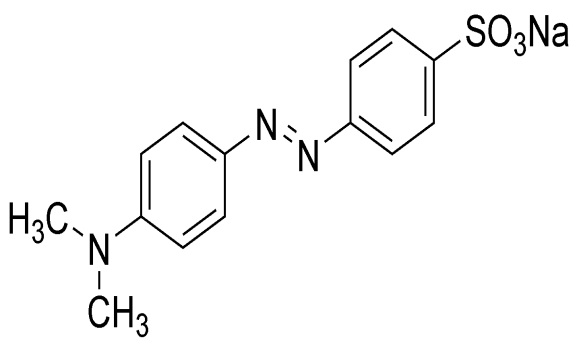
Spectrophotometric Investigation of Redox Reaction between Methyl Orange and Tetraoxomanganate(VII) Ions in Aqueous Acidic Medium: Kinetics and Mechanistic Approach
DOI :
https://doi.org/10.62050/fjst2026.v10.n2.678Mots-clés :
Mechanism, Methyl orange, Redox, PolymerizationRésumé
The spectrophotometric investigation of the redox reaction between methyl orange (here after referred to as MO) and tetraoxomanganate (VII) has been studied in aqueous acid using sodium chloride (NaCl) as an ionic strength I = 0.50 Cmol dm-3, hydrogen ion concentration = 1.0 * 10-3 mol/dm3 (H2SO4) and Temperature = 24±1 0C. The redox reaction showed a stoichiometry of 2:1 and the overall reaction conforms to the rate law: -d[MO]/dt =k2[MO][MnO-4]. The reaction is first order with respect to both the oxidant and reductant concentrations. Lack of polymerization from this reaction suggests probable absence of free radical formation during the course of the reaction. The rate of reaction showed lack of hydrogen ion concentration dependence. The result of ionic strength showed zero effect on the rate of the reaction. Added ions catalyzed the rate of the reaction. Outersphere mechanism is proposed for this reaction.
##plugins.themes.default.displayStats.downloads##
Références
Adetoro, A., Babayo, S. A., Akiode, O. K., Iyun, O. R. and Anene A., (2023). Reaction mechanism between synthesized 2-amino heterocyclic dye with bromate ion in aqueous hydrochloric acid: A kinetic approach. Journal of Chemical Society of Nigeria, 48(1), 192-204.
Anwetin, I. B, Okon, I. E. and Jones, F. (2023). Kinetics and mechanism of oxidation of theophylline by permanganate ion in aqueous sulphuric acid medium. The Pacific Journal of Science and Technology, 24(1), 103-110.
Benson, D. (1969). Mechanism of Inorganic Reactions in Solution. McGraw-Hill, UK, pp. 153-160.
Birk, J. P. (1978). Kinetics and mechanism of the reduction of bromated ion by hexachloroiridate(III). Inorg. Chem., 17, 504 –506.
Dash, S., Patel, S. and Mishra, B. K. (2009). Oxidation by permanganate: synthetic and mechanistic aspects. Tetrahedron, 65(4), 707–739.
Ji, S. (2012). Molecular Theory of the Living Cells: Concepts, Molecular Mechanism and Biomedical Applications. Springer, New York.
Jones, F., Anwetin, I. B., Okon, I. and Andem K. E, (2023). Spectrophotometric study of kinetics of redox reaction between caffeine and permanganate ion in aqueous acidic medium. Asian Journal of Chemical Science, 13(3), 1-9.
Libby, W. F (1952). Theory of electron exchange reaction in aqueous solution. Journal of Physical Chemistry, 56(7), 63-68.
Liu, T. and Shi, C. (2005). Improvement of methyl orange preparation. Guangzhou Chemical Industry, 43(1), 76-77.
Marcus, R. A. (2006). Electron transfer reactions in chemistry: Theory and experiment. Pure and Applied Chemistry, 69(1), 13-29.
Mayer, T. J. and Taube, H. (1987). Comprehensive Coordination Chemistry: The synthesis, Reaction, Properties and Application of Coordination Compounds. Pergamon Press, UK, pp. 331, 357, 384.
Myek, B, Idris, S. O, Onu, A. D. and Yakubu, M. K (2018). Kinetics and mechanism of the oxidation of orange II by permanganate ion in aqueous acidic medium. Nigerian Research Journal of Chemical Sciences, Department of Chemistry, UNN, 5, 98 – 107.
Rao, P. V. S., Murty, P. S. N., Murty, R. S. V. and Murty, B. A. N. (1978). Surface methodology for decolourization of azo dye methyl orange by bacterial consortium: Produced enzymes and metabolites characterization. J. Indian Chem. Soc. Lv, 1280 Chemical Engineering
Reimers, J. R., Mckemmish, L., Mckenzir, R. H. and Hush, N. S. (2005). A unified diabatic description for electron transfer reactions, isomerisation reactions, proton transfer reactions and aromaticity. Physical Chemistry Chemical
Physics, 17(38), 24598-24617.
Reynolds, W. L. and Lumry, R. (1966). Mechanisms of Electron Transfer. The Ronald Press Company, New York, pp. 1-10.
Susan, B. P., Elmars, R. K. and Schatz, P. N. (1978). Vibronic coupling model for calculation of mixed valence absorption profiles. Journal of American Chemical Society, 100(10), 2996-3005.