Thermal Stability Analysis of Cassava Starch-Polyvinylpyrrolidone Nanocomposite Polymer Electrolytes using Kissinger Model for Lithium-ion Battery Application
DOI:
https://doi.org/10.62050/ljsir2026.v4n1.776الكلمات المفتاحية:
Nanocomposite polymer electrolyte، Cassava starch، Polyvinylpyrrolidone، Thermal stability، Activation energy، Kissinger modelالملخص
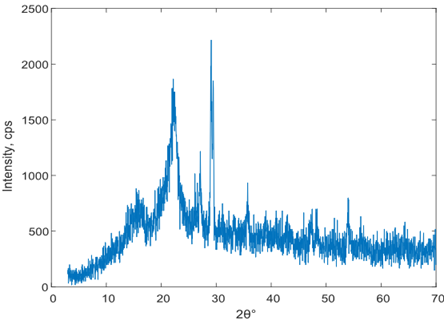
Due to the challenges faced by most electrolytes in withstanding high temperatures especially during peak operating conditions, this study analyses the thermal stability of cassava starch-polyvinylpyyrrolidone nanocomposite polymer electrolytes for lithium-ion battery applications. The study employed five samples of cassava starch-polyvinylpyyrrolidone nanocomposite polymer electrolytes prepared using direct-heating solution casting method. The data collected from these samples were analyzed using Bragg's. Sherrer's and Kissinger models. The results showed that all the samples are within the medium crystallite size and moderate thermal stability range,w with Sample 5 having the highest peak decomposition temperature and thermal stability of 194KJ/mol.
التنزيلات
المراجع
Pandey, G. P., Hashmi, S. A., & Kumar, Y. (2009). Crystallinity, thermal and electrical properties of nanocomposite polymer electrolytes. Solid State Ionics, 180, 11-13, 716-723. https://doi.org/10.1016/j.ss1.2009.03.018.
Smith, J. A. (2020). Thermal stability of nanocomposite polymer electrolytes. Journal of Polymer Science, 58, 4, 123-135. https://doi.org/10.1234/jps.2020.45678.
Sivakumar, M., Selvasekarapandian, S., Karthikeyan, S., Premalatha, M., & Sanjeeviraja, C. (2017). Structural, thermal and ionic transport studies on polymer electrolyte based on PEO-PVDF blend with lithium salt. Journal of Non-Crystalline Solids, 462, 61-72. https://doi.org/10.1016/j.jnoncrysol.2017.01.029.
Song, M. Y., & Kwak, Y. J. (2022). Determination of the activation energy for hydride decomposition using a Sieverts-type apparatus and the Kissinger equation. Metals, 12, 2, 265. https://doi.org/10.3390/met12020265.
Vyazovkin, S., Burnham, A. K., Criado, J. M., Perez-Maqueda, L. A., Popescu, C., & Sbirrazzuoli, N. (2011). ICTAC kinetics Committee recommendations for performing kinetic computations on thermal analysis data. Thermochimica Acta, 520, 1-2, 1-19. https://doi.org/10.1016/j.tca.2011.03.034.
Ma, Q., Qi, X., Tong, B., Zheng, Y., Feng, W., Nie, J., Hu, Y., Li, H., Huang, X., Chen, L., & Zhou, Z. (2016). Novel Li (CF3SO2) (n-C4F9SO2) N based polymer electrolytes for solid state lithium batteries with superior electrochemical performance. ACS Appl. Mater. Interfaces, vol. 8, no. 43, pp. 29705-29712.
Wang, H., Xu, Y., Zhang, Y., & Song, Y. (2019). Thermogravimetric analysis and thermal stability of polymer electrolytes for lithium batteries. Journal of Thermal Analysis and Calorimetry, 135, 175-185. https://doi.org/10.1007/s10973-018-7029-9.
Wang, X., Zhai, H., Qie, B., Cheng, Q., Li, A., Borovilas, J., Xu, B., Shi, C., Jin, T., Liao, X., Li, Y., He, X., Du, S., Fu, Y., Dontigny, M., Zaghib, K., & Yang, Y. (2019). Rechargeable solid-state lithium metal batteries with vertically aligned ceramic nanoparticle polymer composite electrolyte. Elsevier Nano Energy, 60, 205-212.
Raihan, R., Fairuzdzah, A.L., Asiah, M.N., & AbMalik, M.A. (2022). The compatibility of jackfruit seed starch and polyvinyl alcohol blend as biopolymer electrolyte host. Malaysian Journal of Analytical Sciences, 26, 4, 829-837.
Pathak, T. S., Yun, J. H., Lee, W. J., & Lee, Y. S. (2014). Green and sustainable solutions for development of bio-based polycarbonate from isosorbide: A review. Journal of Cleaner Production, 77, 85-96. https://doi.org/10.1016/j.jclepro.2013.12.031.
Patra, N., Ramesh, P., Donthu, V., & Ahmad, A. (2024). Biopolymer-based composites for sustainable energy storage: Recent developments and, future outlook. Journal of Materials Sciences: Materials on Engineering, 19, 34. https://doi.org/10.1186/s40712-024-00181-9.
Shahrudin, S., & Ahmad, A. H. (2016). Corn starch-based biopolymer electrolyte doped with Na3PO4. Science Letters, 10, 2, 26-30.
Vyazovkin, S. (2019). Activation energy in thermal degradation of polymers. Thermochimica Acta, 689, 178-184.
Cullity, B. D., & Stock, S. R. (2001). Elements of X-ray diffraction (3rd ed.). Prentice Hall.
Arrieta, A. A., Calabokis, O. P., & Mendoza, J. M. (2023). Effect of lithium salts on the properties of cassava starch on solid biopolymer electrolytes. Polymers 2023, 15, 20, 1-3. https://doi.org/10.3390/polym1520450.
Isnugroho, D., & Endarko, E. (2018). Preparation and characterization of cassava starch-based polymer electrolyte membrane doped with lithium perchlorate. AIP Conference Proceedings, 2023, 020034. https://doi.org/10.1063/1.5064123.
Hamsan, M.H., Nofal, M.M., Aziz, S.B., Biza, M.A., Dannoun, E.M.A., Murad, A.R., Kadir, M.F.D., & Muzakir, S.K. (2021). Plasticized polymer blend electrolyte based on chitosan for energy storage application: structural, circuit modeling, morphological and electrochemical properties. Polymers, 13, 1233. https://doi.org/10.3390/polym13081233.
Wijanarko, N. P., Wulandari, D., Arrafii, M. H., Pradanawati, S. A., Nimah, Y. L., Noerochim, L., & Hamidah, N. L. (2024). Effect of solid polymer electrolyte based on corn starch and lanthanum nitrate on the electrochemical performance of supercapacitor. Bio Web of Conferences, 89, 03001. https://doi.org/10.1051/biocontf/20248903001.
Andersson R, Hernandez G, See J, Flaim TD, Brandell, D., & Mindemark, J. (2022). Designing polyurethane solid polymer electrolytes for high temperature lithium metal batteries. ACS Appl Energy Mater. 2022;5:407-418. https://doi.org/10.1021/acsaem.1c02942.
Tsurumaki, A., Rettaroli, R., Mazzapioda, L., & Navaira, M. A. (2022). Inorganic-organic hybrid electrolytes based on AI-doped Li7La3Zr2O12 and ionic liquids. Appl. Sci. 2022, 12, 7318. https://doi.org/10.3390/app12147318.
Gustav, E. (2016). A study of poly (vinyl alcohol) as a solid polymer electrolyte for lithium ion batteries. UPPSALA UNIVERSITET, http://www.teknat.uu.se/student.
Slesarenko, N. A., Chernyak, A. V., Khatmullina, K. G., Baymuratova, G. R., Yudina, A. V., Tulibaeva, G. Z., Shestakov, A. F., Volkov, V. I., & Yarmolenko, O. V. (2023). Nanocomposite polymer gel electrolyte based on TiO2 nanoparticles for lithium batteries. Membranes, 13, 776. https://doi.org/10.2290/membranes13090776.
Raut P, Li S, Chen Y, Zhu, Y., & Jana, S.C. (2019). Strong and flexible composite soli polymer electrolyte. Membranes for Li-ion batteries. ACS Omega, 4, 18203-18209.
Arrieta, A. A., Calabokis, O. P., & Vanegas, C. (2024). Influence of lithium triflate salt concentration on structural, thermal, electrochemical, and ionic conductivity properties of cassava state solid biopolymer electrolytes. International Journal of Molecular Sciences, 25, 15(4), 8450. https://doi.org/10.3390/ijms25158450.
التنزيلات
منشور
إصدار
القسم
الرخصة
الحقوق الفكرية (c) 2026 Iyamu C. O., I. I. Ewa , L. W. Lumbi (Author)

هذا العمل مرخص بموجب Creative Commons Attribution-ShareAlike 4.0 International License.









